Moravek manufactures GMP carbon-14 labeled APIs under a Drug Manufacturing License issued by the California Department of Public Health – Food and Drug Branch following an audit of our Quality System.

GMP production is carried out in one of four cleanrooms which are annually certified to be ISO Class 7 compliant.

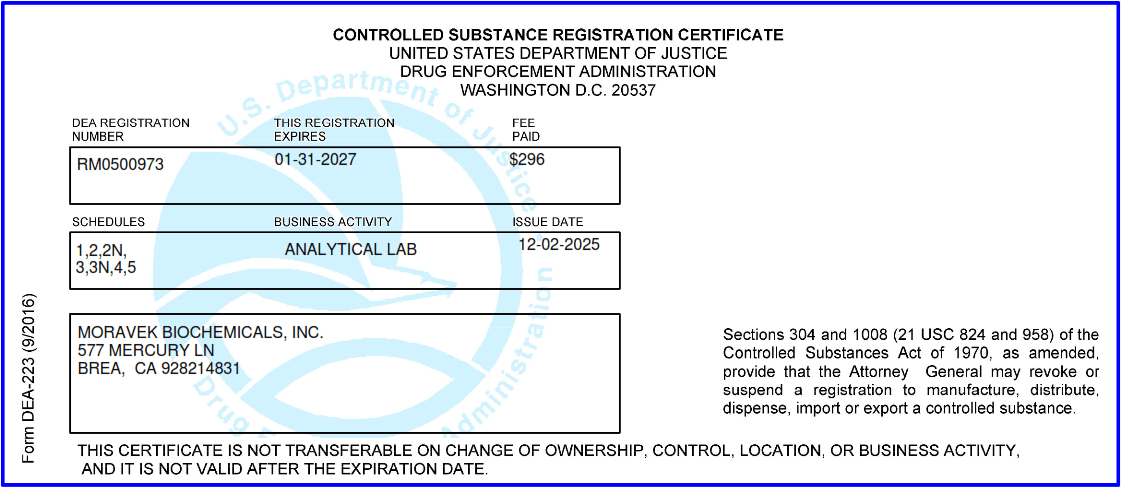
Moravek also has a license from the DEA to handle the full range of Schedule I – V controlled substances.
Moravek handles radioactive materials under a Radiological Health Certificate from the California Department of Public Health – Radiologic Health Branch.

Choose Moravek as your supplier for custom labeled compounds!
Contact our team now to get started or request a quote.
